According to the World Health Organisation (WHO), counterfeit medicines are found everywhere in the world. They include mixtures of toxic substances, and inactive, ineffective preparations; 700,000 people die annually from fake malaria and tuberculosis drugs alone. The WHO also reports that the annual earnings from substandard or counterfeit drugs stand at about $200 billion USD.
Many innovative solutions are being proposed to help tackle the spread of counterfeit drugs in developing countries.
Technological innovations to track counterfeit drugs
Ashifi Gogo from Ghana is an entrepreneur with an innovative solution involving mobile phones. In simplified terms, the idea is to place a scratch-off label on the products; the label contains a unique code, which the consumer sends via SMS and then receives a reply as to whether the product is authentic or not.
In this TEDx Boston talk, Ashifi Gogo explains how his solution works by combining cell phones, community, and the cooperation of governments and pharmaceutical companies:
After the Duo-Cotecxin incident in Kenya, Holley-Cotec, the company that manufactures the antimalarial, introduced a new technology to make the tablets tamper-proof. James Nyikal, director of Medical Services in Kenya explains :
New packets of the drugs will have three-dimensional hologram seals and other features to indicate the drugs are authentic.

“Is this drug real or counterfeit? SMS text with Pharma Secure to find out.” Image by Wayan Vota on Flickr (CC BY-NC-SA 2.0)
Rajendrani Mukhopadhyay reviews the solutions currently available to identify genuine medicines. He shares some surprising anecdotes:
The screening first consists of a close visual inspection. Inspectors scrutinize the packages, because they need to be certain that an intention exists to deceive the consumer. “If you just have the tablets or the capsules, even if they have no active ingredient, it still could have been an innocent mistake in the factory. […] Some of the packaging errors on counterfeit drugs are quite comical. On one, the expiry date was before the manufacturing date, so we knew something wasn’t quite right!”
He also mentions the challenges of screening in poor settings:
Challenges in developing countries mean that investigators have to think creatively and come up with effective analytical techniques that cost pennies and are robust. “We found out that many of the fake artesunates, posing as a particular brand produced in China, contained calcium carbonate,” says Green [Michael Green from the U.S. Centers for Disease Control and Prevention]. Since the authentic brand did not contain calcium carbonate, he and his colleagues were able to distinguish counterfeits from the genuine tablets by adding vinegar to bits of the tablets.
A growing challenge for the developing world
More important than their economic impact, counterfeit medicines pose a significant global public health problem. Not only do they endanger the lives and the well-being of patients, they also undermine confidence in healthcare systems and health professionals.
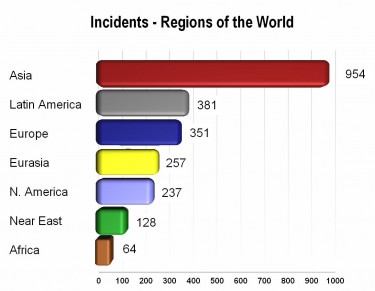
The following chart by the Pharmaceutical Security Institute (PSI) details the geographical distribution of confirmed counterfeiting incidents in 2011; the issue is increasingly challenging to tackle for the developing world because of the growing volume of fake drugs and the required infrastructure.
To the point, PSI notes:
The regions that are more frequently linked to incidents are not necessarily those with weak enforcement and inspection programs. Rather, countries in these regions are effectively identifying pharmaceutical crime through law enforcement activity and inspections by drug regulatory agencies.
Despite the innovation mentioned earlier, detecting counterfeit drugs is still a very difficult task and requires both advanced technology and the capacity to strictly enforce international regulations.
For instance, many defective and counterfeit antimalarials were reported in developing countries [fr] this year but only after patients had been treated. In 2012, a study showed that one-third of antimalarial medications in Southeast Asia and Sub-Saharan Africa failed chemical analysis, packaging analysis, or were falsified. In 2007, the Ministry of Health in Kenya issued a warning about counterfeit antimalarial medication Duo-Cotecxin being sold in Nairobi and how to detect it.
Lawrence Evans, a researcher who has conducted a study in Guyana and Suriname on the quality of antimalarial drugs says:
Besides the presence of medicines not included in the World Health Organization malaria treatment guidelines, the ease with which medications were procured without accurate diagnosis poses another major risk to patients’ safety. In addition, this could have serious implications for the development of drug-resistant strains of Plasmodium parasites, particularly Falciparum, as novel treatments are not foreseeable in the near future.
Prisca in Madagascar explains [fr] why people do not go to pharmacies anymore and venture into the informal market:
des personnes n'hésitent plus à revendre des produits pharmaceutiques sans ordonnance. Selon Fara, propriétaire d'un commerce prospère ” Les médicaments que nous vendons sont les mêmes qu'en pharmacie, puisque je les achète chez un grossiste agréé. Mais comme je n'ai pas autant de charges qu'une pharmacie, ils sont deux fois moins chers “. Le problème des consommateurs est donc le prix.
While it is important to determine the circuit of counterfeit drugs to fight efficiently against this trafficking, many countries warn that this cannot overshadow the need to innovate in other areas as well to fight diseases. Henry Neondo writes for African Science News that malaria must be fought more effectively in Kenya, including the search for a vaccine:
Kenya has been selected among nine other African countries that will benefit from a new multi-billion shillings drive in search for an anti-malaria vaccine. [..] “New malaria drugs and a vaccine are urgently needed in Africa, where malaria has grown resistant to the cheapest and most widely-used treatments. As several promising new drugs and vaccines move through the research pipeline, there is a need to build African capacity to conduct large-scale clinical trials of these drugs and vaccines over the next decade,” said Prof Binka [Indepth Network Executive Director].







4 comments
Tracking Counterfeit Medicines in the Developing World · Global Voices…
Great summary article on some current innovations being implemented to tackle the counterfeit medicine industry worldwide….
There is very little evidence of widespread counterfeiting of drugs in developing countries, as counterfeiting is normally defined, but plenty of evidence of that substandard drugs are an important health problem. Since the problems of low quality drugs and counterfeiting are really different, many object to lumping the categories together. In any event, the $200 billion number has no basis at all.
Good research should have given due credit to the originator of the so-called sms tracking idea. He is from Ghana but he is not the one in the picture.
How can we combat Anti-Counterfeiting?
My company, Recunnect Ltd are organising an event called
‘Anti-Counterfeiting Pharma 2015’ on 11th & 12th March 2015 at Marriott
Hotel, Regents Park, London, UK.
It is a senior networking event which encourages you to learn about
the current trends in anti-counterfeiting, brand protection and security
packaging. Identifying the key strategies for combating anti-counterfeiting,
online pharmaceutical crime, products and latest solutions.
‘Anti-Counterfeiting Pharmaceutical Conference’ will bring experts
from cross-sector organisations together to share their knowledge and
experience on the key issues and latest technological developments. The aim is
to foster discussion on how to tackle counterfeiting and improve patient
safety.
For more information, please visit our website-
http://www.recunnect.com/-anti-counterfeiting-pharma-2015.html. To get in touch
with us please email us on jasneet@recunnect.net or call +44 (0)
2071129183 / +44(0)7712128928. For registrations, please call us and quote the account
code RES001 when making a booking. You can also follow our event on
Facebook-https://www.facebook.com/events/1599475776942677/?ref_dashboard_filter=upcoming
Twitter-https://twitter.com/pharma_anti &
Linkedin-https://www.linkedin.com/company/anti-counterfeiting-pharma-2015?trk=biz-companies-cym